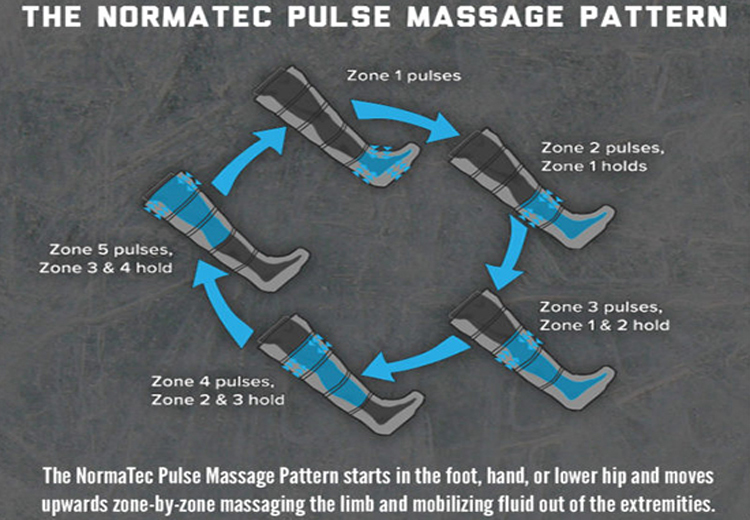
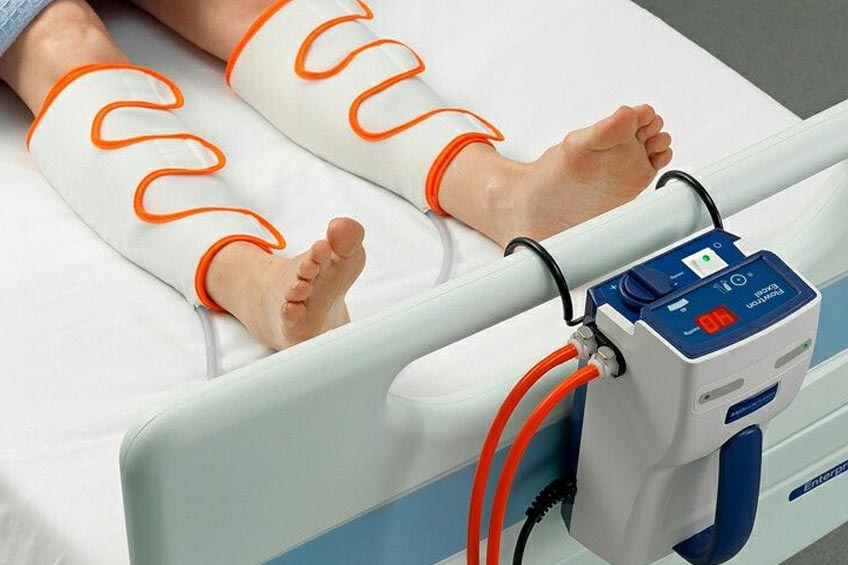
The DMEMACs released an updated Local Coverage Determination (LCD) revision that eliminates the requirement of serial four week trials of conservative therapy in order to obtain a Pneumatic Compression Device (PCD).

Philip Miziniak II posted on LinkedIn

Andrea Danielle Stanley, MSN AGPCNP-C CWON-AP DAPWCA on LinkedIn: Medicare Removes the Requirement That a Patient Fail Four Weeks of…

Kimberly Weatherly, BSN, RN, CWOCN on LinkedIn: Two great companies partnering to elevate wound care standards. Exciting!

Fight the Good Fight Archives - Caroline Fife M.D.

Philip Miziniak II posted on LinkedIn

FinalProgram

Andrea Cooper Neal, MOT, OTR/L, CLT-LANA on LinkedIn: Nashville Breast Center had fun supporting the amazing Survivor Fitness…

Philip Miziniak II posted on LinkedIn

David Schrader on LinkedIn: #healogics #ths #partnership #patientjourney

Kimberly Weatherly, BSN, RN, CWOCN posted on LinkedIn

More of Your Letters: Skin Substitutes, CMS, Failed Proposals and the Broken System - Caroline Fife M.D.

Betty Granado BSN, RN, CCM on LinkedIn: Medicare Removes the

Andrea Danielle Stanley, MSN AGPCNP-C CWON-AP DAPWCA on LinkedIn: Medicare Removes the Requirement That a Patient Fail Four Weeks of…

Philip Miziniak II on LinkedIn: Nerve Block Shows Promise for Long COVID-Related Olfactory or Gustatory…

Raeanna Smith on LinkedIn: Medicare Removes the Requirement That a Patient Fail Four Weeks of…