20.If Z is a compressibility factor, van der Waals equation at low pressure can be written as
20-If Z is a compressibility factor- van der Waals equation at low pressure can be written as
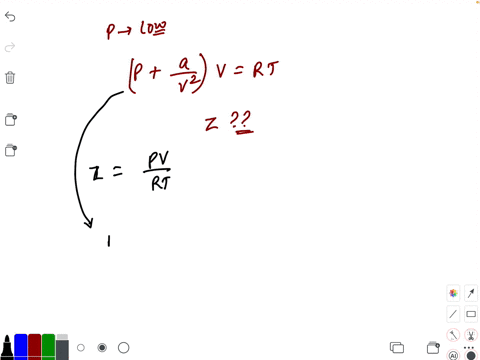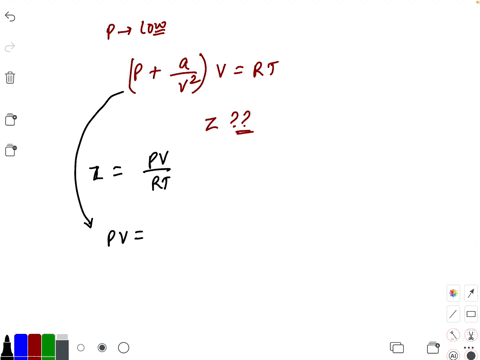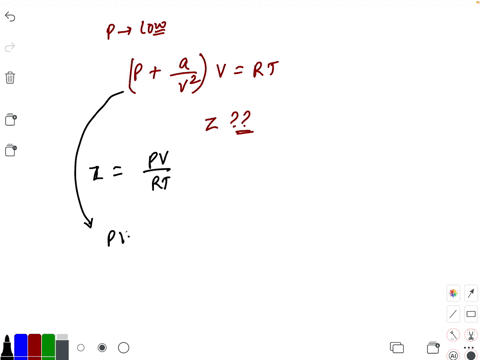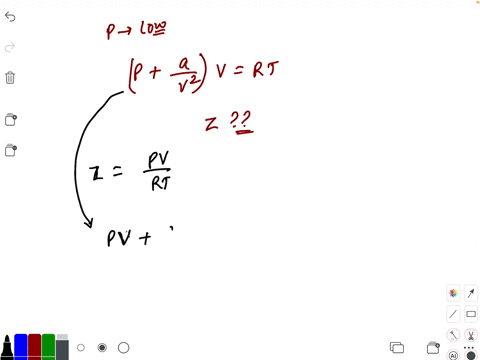
⏩SOLVED:At low pressures, van der Waals' equation is written as…

⏩SOLVED:For a van der Waals gas with given values of a and b,…

Which of these are correct? A) Z, compressibility factor, low pressure can be written as z = B) Z, low pressure can be written as z = 1 + P C) Z

Bengali] What will the value of compressibility factor (Z) be for a g
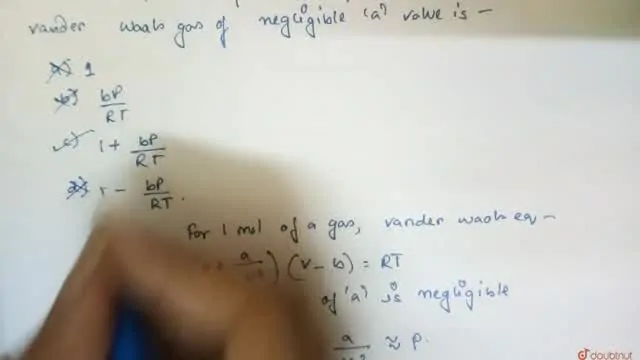
Bengali] The compresibility factor (Z) of one mole of a van der waals

Cubic equations of state - Wikipedia

If Z is a compressibility factor, vander Waals equation low pressure can be written as [JEEN (0)2=1 Rang (1) Z= 1 + RT Pb (2) Z 2)2=1= = 1 - 2= (3) Z = 1 - 42=1 (4)Z = 1 + VRT

Compressibility factor (Z) for a van der Waals real gas at critical po

a) Compressibility factor Z obtained from the Lee-Kesler EoS, and
The compressibility factor for one mol of a vanderwalls gas at 0 degree c and 100atm pressure is .5 then what will be the volume of 2 mols of this gas
Gas Compressibility Factor and Control Valve Sizing

If Z is a compressibility factor, Van der Waals equation at low pressure can be written as

plotting - How to plot Compressibility factor Z vs Pressure P using ParametricPlot? - Mathematica Stack Exchange

If Z is a compressibility factor, van der Waals equation at low pressure can be written as [JEE

At low pressure the van der Waals' equation is reduced to [P +(a)/(V^(







