

The compression factor (compressibility factor) one mole of a van der Waals gas 0°C and 100 atm pressure is found to be 0.5. Assuming that the volume of a gas molecule is
Why can't anymore atoms enter in the excluded volume (region of 2 atoms) in the volume correction given by van der Waals equation? - Quora
The compression factor (compressibility factor) for one mole of a van der Waals' gas - Sarthaks eConnect
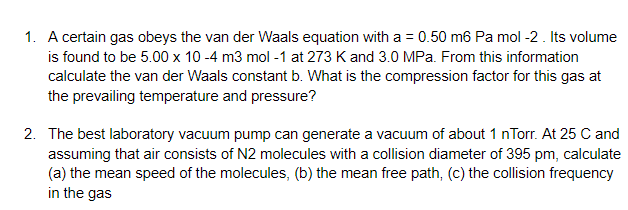
Solved A certain gas obeys the van der Waals equation with a

Kirkwood–Buff-Derived Force Field for Peptides and Proteins: Applications of KBFF20

Solved At high gas densities, the van der Waals equation der

The compression factor (compressibility factor) one mole of a van der Waals gas 0°C and 100 atm pressure is found to be 0.5. Assuming that the volume of a gas molecule is
How do Van der Waals constants a and b depend on temperature, pressure and volume? - Quora

Solved 2. Fugacity for a van-der-Waals gas Let's get a feel

1148 questions with answers in GAS

The compression factor compressibility factor for 1 mole of a van der Waals' gas at 0∘ C and 100 atmospheric pressure is found to be 0.5 . Assuming that the volume of

the compression factor one mole of a vander waals gas 0 C and 100 atm pressure is found to be 0.5

Chemical Process Dynamics and Controls, 2018a

What is the compressibility factor (Z) for 0.02 mole of a van der Waal

the compression factor one mole of a vander waals gas 0 C and 100 atm pressure is found to be 0.5







